Category Archives: Infectious Diseases
PROPHY-VAP Trial
Study To Review
One Sentence Summary
The PROPHY-VAP trial showed that a single dose of ceftriaxone can significantly reduce early-onset VAP in mechanically ventilated brain-injured patients, offering a potential change in clinical practice for VAP prevention.
Introduction
Methods
Results
Ventilator-associated pneumonia (VAP) is a significant concern in intensive care units (ICU), especially prevalent among brain-injured patients who are mechanically ventilated. With VAP incidence rates ranging from 22% to 71%, depending on the study, and brain injury being a notable independent risk factor, the prevention of this infection is paramount. Current standard of care includes a set of measures aiming to reduce the incidence of VAP; however, the challenge of early versus late onset VAP and the associated risk of multi-drug resistant organisms necessitate ongoing research and innovation in prophylactic strategies. The PROPHY-VAP trial is a pivotal study that explores the use of prophylactic antibiotics, specifically ceftriaxone, to prevent early-onset VAP in brain-injured patients. By addressing the controversial cut-off for early VAP onset in this population and considering the unique susceptibility profile of their infections, this trial holds significant importance for enhancing patient outcomes and refining ICU protocols.
PROPHY-VAP was a multicentre, randomized, double-blind, placebo-controlled trial conducted in nine French ICUs. The study enrolled comatose adult patients (Glasgow Coma Scale score ≤12) requiring mechanical ventilation post-acute brain injury. Patients were randomly assigned to receive either intravenous ceftriaxone 2 g or a placebo within 12 hours of tracheal intubation. The primary outcome was the incidence of early VAP (2nd to 7th day of mechanical ventilation). The study excluded patients with high death risk within 48 hours, previous hospitalizations for coma, contraindications to beta-lactams, and those receiving antibiotics for pre-existing infections.
- The PROPHY-VAP trial was conducted over a period spanning from October 14, 2015, to May 27, 2020, during which 345 patients were randomized in a 1:1 ratio to receive either ceftriaxone (171 patients) or placebo (174 patients). The study observed a marked reduction in the incidence of early VAP in the ceftriaxone group, with only 14% (23 patients) developing the infection compared to 32% (51 patients) in the placebo group, translating to a hazard ratio of 0.60. This significant decrease in early-onset VAP incidence, demonstrated in a double-blind, placebo-controlled setting, suggests that a single dose of ceftriaxone can be an effective prophylactic intervention in brain-injured patients requiring mechanical ventilation.
A detailed analysis of the patient demographics within the trial revealed that out of the 319 patients included in the analysis, 166 were men and 153 were women. Adjudication confirmed a total of 93 cases of VAP, of which 74 were early infections. Importantly, the study reported no increase in multi-drug resistant organisms or adverse effects attributable to the administration of ceftriaxone. These results indicate that the intervention was not only efficacious in reducing the incidence of early VAP but also safe for the patients, with no additional risk of fostering antibiotic resistance. The authors report that these findings provide a strong argument for the inclusion of a single dose of ceftriaxone in VAP prevention bundles for this patient population.
Conclusion
The study demonstrates that a single dose of ceftriaxone significantly reduces the risk of early VAP in brain-injured patients requiring mechanical ventilation, without adverse microbiological consequences. This finding supports the inclusion of an early ceftriaxone dose in VAP prevention protocols for brain-injured patients.
Impression
The PROPHY-VAP trial’s findings have sparked debate within the medical community, with practitioners expressing both interest in the potential benefits of ceftriaxone prophylaxis and concern regarding the broader implications of antibiotic use, such as the risk of over-diagnosis of VAP and the absence of data on late-onset VAP and resistant pathogens. These discussions underscore the necessity for further research and careful consideration before integrating these results into standard practice.
Further Readings
Piperacillin-tazobactam plus Vancomycin and Acute Kidney Injury by Caroline Rosario
Introduction
- Vancomycin and piperacillin-tazobactam are combined for broad-spectrum antibiotic coverage including MRSA and Pseudomonas in hospitalized patients.
- AKI, often as acute tubular necrosis, is a known complication of vancomycin, especially with higher doses and co-administration of nephrotoxic drugs.
- Piperacillin-tazobactam alone has minimal nephrotoxicity (<1%); its nephrotoxicity is usually due to acute interstitial nephritis.
- Reported AKI rates vary in literature based on AKI definition and target population.
- Both drugs affect OAT1/3 transporters in the kidney, which are crucial for creatinine clearance and are especially significant in patients with CKD.
Pharmacology
| Vancomycin | Piperacillin-tazobactam4 | |
| Dose | Depends on infection and PK/PD target General dosing for systemic infections: IV 15-20 mg/kg IV Q8-12H for systemic infections | Standard infusion: 3.375 g IV Q6H over 30 minAntipseudomonal: 4.5 g IV Q6-8H over 30 minExtended infusion: 4.5 g IV then 3.375-4.5 g over 4 hours Q8H |
| Administration | Administer IV over ≥60 minutes at concentrations ≤5 mg/mL to reduce the risk of vancomycin infusion reaction | Standard infusion: Infuse over 30 min Extended infusion: Infuse loading dose over 30 min, start maintenance dose four hours later infused over 4 hours |
| PK/PD | Negligible oral bioavailability T1/2 = 4-6 hours Renally eliminated (40-100% unchanged) AUC:MIC dependent kinetics, PK/PD target AUC/MIC ≥400 µg/mL; surrogate serum trough concentrations often used | T1/2 = 0.7-1.2 hours Renally eliminated (80% unchanged) Dose adjust at CrCl<40 T>MIC dependent kinetics, prolonged infusions enhance efficacy |
| Adverse Effects | Nephrotoxicity Ototoxicity Vancomycin-infusion reaction (flushing, hypotension, tachycardia) | GI upset (diarrhea, nausea, constipation) Headache Rash, pruritis |
| Drug Interactions and warnings | Substrate of OAT1/3 +/- Inducer of OAT1/3 ↑ nephrotoxicity: aminoglycosides, aspirin | Piperacillin: substrate and inhibitor of OAT1/3∆, Tazobactam: substrate of OAT1/3 Interactions: Probenecid (↑ piperacillin-tazobactam), Methotrexate (↑ methotrexate) |
| Compatibility | Compatible with dextrose, NS, LR Incompatible with lipid emulsion | LR: only the formulation containing EDTA is compatible for Y-site administration Not chemically stable in solutions containing sodium bicarbonate or solutions that significantly alter pH Cannot be added to blood products or albumin hydrolysates |
| Comments | Serum troughs are a poor proxy of 24-hour AUC, trough-guided regimens have been shown to exceed the target AUC in 60% of adults10 | Useful in the ED for anaerobic coverage in Grade III open fractures, pneumonia with lung abscess or empyema, and empiric antipseudomonal coverage in patients with risk factors |
Overview of Evidence
| Author, year | Design/ sample size | Intervention & Comparison | AKI definition | Outcome |
| Sanz et al., 2002 | Prospective, multi-center (n = 969) | Amikacin+cefepime vs. amikacin+piperacillin-tazobactam | ↑ SCr ≥50% from baseline | No difference in severe nephrotoxicity between amikacin+piperacillin-tazobactam vs. amikacin+cefepime |
| Karino et al., 2016 | Retrospective cohort and nested case-control studies (n = 320) | Vancomycin+piperacillin-tazobactam standard infusion vs. Vancomycin+piperacillin-tazobactam extended-infusion | RIFILE criteriaAKIN criteriaVancomycin consensus guideline definition | AKI occurred in 33% of patients receiving vancomycin+piperacillin-tazobactamUse of extended infusion piperacillin-tazobactam did not increase risk of AKI Highest daily incidence of AKI occurred on day 5 of combination therapy |
| Hammond et al., 2017 | Meta-analysis of 14 observational studies (n = 3549) | Vancomycin+piperacillin-tazobactam vs. vancomycin+any β-lactam or vancomycin alone | All included studies used one of the following: RIFLE criteriaAKIN criteria↑ SCr ≥100% or >0.5 mg/dL | Vancomycin+piperacillin-tazobactam greater association with AKI (aOR, 3.11; 95% CI, 1.77–5.47) Highest incidence of AKI in patients admitted to the ICU (OR 3.83 95% CI, 1.67-8.78) |
| Rutter et al., 2017 | Retrospective matched cohort (n = 4103) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | RIFLE criteria | Vancomycin+piperacillin-tazobactam 2.18 times more likely to cause AKI vs. vancomycin+cefepime (95% CI, 1.64–2.94) Vancomycin doses between 3 and 4 g daily used, |
| Peyko et al., 2017 | Prospective observational cohort (n = 85) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime or vancomycin+meropenem | KDIGO | Incidence of AKI was higher in with vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime or meropenem (37.3% vs. 7.7% P = .005) |
| Rutter and Burgess et al., 2017 | Retrospective matched cohort (n = 2448) | Vancomycin+piperacillin-tazobactam vs. Vancomycin+ampicillin-sulbactam | RIFLE criteria | Increased risk of AKI with vancomycin+piperacillin-tazobactam (aOR, 1.77; 95% CI, 1.26–2.46), no increased rate of AKI with vancomycin+ampicillin-sulbactamRates of AKI similar for piperacillin-tazobactam and ampicillin-sulbactam without vancomycin |
| Jeon et al., 2017 | Retrospective matched cohort (n = 5335) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | ↑ SCr ≥0.3 mg/dL or ≥50% from baseline | Vancomycin+piperacillin-tazobactam associated with a higher risk of AKI vs. vancomycin-cefepime (aHR, 1.25; 95% CI, 1.11–1.42.) |
| Mousavi et al., 2017 | Retrospective matched cohort (n = 280) | Vancomycin+piperacillin-tazobactam standard infusion vs. Vancomycin+piperacillin-tazobactam extended-infusion | RIFLE criteriaAKIN criteria | Similar rate of AKI between vancomycin+piperacillin-tazobactam standard infusion vs. vancomycin+piperacillin-tazobactam extended-infusionHigher vancomycin troughs were observed in the extended infusion group |
| Miano et al., 2022 | Prospective, observational | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime for ≥48 hours | ↑ SCr vs. ↑ Cystatin C vs. ↑ BUN | Vancomycin + piperacillin-tazobactam ➡️ ↑ serum creatinine-defined AKI, but no change in cystatin C, BUN, or AKI outcomes (dialysis/mortality).Indicates vancomycin + piperacillin-tazobactam AKI may be pseudotoxicity. |
| Qian et al, 2023 (ACORN Trial) | Randomized controlled Trial N=2511 | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | KDIGO ↑ SCr ≥0.3 mg/dL or ≥50% from baseline | The highest stage of acute kidney injury or death was not significantly different between the cefepime group and the piperacillin-tazobactam groupThe incidence of major adverse kidney events at day 14 did not differ between groups (124 patients [10.2%] in the cefepime group vs 114 patients [8.8%] in the piperacillintazobactam group~77% of each concurrently received vancomycin |
RIFLE, AKIN and KDIGO definitions of AKI are based upon ↑ in serum creatinine or ↓ in urine output
Conclusions
- Since 2011, evidence indicates combined vancomycin+ piperacillin-tazobactam may be nephrotoxic.
- Most studies were retrospective, defining nephrotoxicity by creatinine-based AKI.
- Recent data show this AKI definition doesn’t align with severe AKI outcomes (hemodialysis/mortality).
- Non-tubular secretion biomarkers (Cystatin C, BUN) didn’t show the same AKI increase.
- Despite >50 studies linking the drug combo with AKI, some expert report true renal risk is likely minimal.
- In emergencies, timely antibiotic use is vital; nephrotoxicity concerns shouldn’t delay this combo, especially for short use.
References
- Micromedex [Electronic version].Greenwood Village, CO: Truven Health Analytics. Retrieved October 4, 2023, from http://www.micromedexsolutions.com/
- VANCOMYCIN HYDROCHLORIDE [package insert]. Rockford, IL: Mylan Institutional LLC; Jul, 2018.
- Blair M, Côté JM, Cotter A, Lynch B, Redahan L, Murray PT. Nephrotoxicity from Vancomycin Combined with Piperacillin-Tazobactam: A Comprehensive Review. Am J Nephrol. 2021;52(2):85-97. doi: 10.1159/000513742.
- Pill MW, O’Neill CV, Chapman MM, Singh AK. Suspected acute interstitial nephritis induced by piperacillin-tazobactam. Pharmacotherapy. 1997 Jan-Feb;17(1):166-9..
- Li H, Yang Q, Gui M, Ding L, Yang L, Sun H, Li Z. Changes of renal transporters in the kinetic process of VCM-induced nephrotoxicity in mice. Toxicol Res (Camb). 2021 Jun 9;10(4):687-695. doi: 10.1093/toxres/tfab048. PMID: 34484661; PMCID: PMC8403606.
- Giuliano CA, Patel CR, Kale-Pradhan PB. Is the Combination of Piperacillin-Tazobactam and Vancomycin Associated with Development of Acute Kidney Injury? A Meta-analysis. Pharmacotherapy. 2016 Dec;36(12):1217-1228. doi: 10.1002/phar.1851.
- Boucher, H. (2023) Piperacillin-tazobactam, Sanford Guide Web Edition. Available at: https://webedition.sanfordguide.com/en/drug-information/antibacterial-agents/penicillins/anti-pseudomonal-penicillins/piperacillin-tazobactam (Accessed: 12 October 2023).
- Yang S, Liu Z, Wang C, Wen S, Meng Q, Huo X, Sun H, Ma X, Peng J, He Z, Liu K. Piperacillin enhances the inhibitory effect of tazobactam on β-lactamase through inhibition of organic anion transporter 1/3 in rats. Asian J Pharm Sci. 2019 Nov;14(6):677-686. doi: 10.1016/j.ajps.2018.11.003.
- Landersdorfer CB, Kirkpatrick CM, Kinzig M, Bulitta JB, Holzgrabe U, Sörgel F. Inhibition of flucloxacillin tubular renal secretion by piperacillin. Br J Clin Pharmacol. 2008 Nov;66(5):648-59. doi: 10.1111/j.1365-2125.2008.03266.x.
- Neely MN, Youn G, Jones B, Jelliffe RW, Drusano GL, Rodvold KA, Lodise TP. Are vancomycin trough concentrations adequate for optimal dosing? Antimicrob Agents Chemother. 2014;58(1):309-16. doi: 10.1128/AAC.01653-13.
- Alvarez-Arango S, Ogunwole SM, Sequist TD, Burk CM, Blumenthal KG. Vancomycin Infusion Reaction – Moving beyond “Red Man Syndrome”. N Engl J Med. 2021 Apr 8;384(14):1283-1286. doi: 10.1056/NEJMp2031891. Epub 2021 Apr 3.
- Vallon V, Eraly SA, Rao SR, Gerasimova M, Rose M, Nagle M, Anzai N, Smith T, Sharma K, Nigam SK, Rieg T. A role for the organic anion transporter OAT3 in renal creatinine secretion in mice. Am J Physiol Renal Physiol. 2012 May 15;302(10):F1293-9. doi: 10.1152/ajprenal.00013.2012. Epub 2012 Feb 15. PMID: 22338083; PMCID: PMC3362066.
- Sanz MA, López J, Lahuerta JJ, Rovira M, Batlle M, Pérez C, Vázquez L, Julià A, Palau J, Gutiérrez M, Capote FJ, Ramos F, Benlloch L, Larrea L, Jarque I; Spanish PETHEMA Group. Cefepime plus amikacin versus piperacillin-tazobactam plus amikacin for initial antibiotic therapy in haematology patients with febrile neutropenia: results of an open, randomized, multicentre trial. J Antimicrob Chemother. 2002 Jul;50(1):79-88. doi: 10.1093/jac/dkf087. PMID: 12096010.
- Watkins RR, Deresinski S. Increasing Evidence of the Nephrotoxicity of Piperacillin/Tazobactam and Vancomycin Combination Therapy-What Is the Clinician to Do? Clin Infect Dis. 2017 Nov 29;65(12):2137-2143. doi: 10.1093/cid/cix675.
- Karino S, Kaye KS, Navalkele B, Nishan B, Salim M, Solanki S, Pervaiz A, Tashtoush N, Shaikh H, Koppula S, Martin ET, Mynatt RP, Murray KP, Rybak MJ, Pogue JM. Epidemiology of Acute Kidney Injury among Patients Receiving Concomitant Vancomycin and Piperacillin-Tazobactam: Opportunities for Antimicrobial Stewardship. Antimicrob Agents Chemother. 2016 May 23;60(6):3743-50. doi: 10.1128/AAC.03011-15.
- Hammond DA, Smith MN, Li C, Hayes SM, Lusardi K, Bookstaver PB. Systematic Review and Meta-Analysis of Acute Kidney Injury Associated with Concomitant Vancomycin and Piperacillin/tazobactam. Clin Infect Dis. 2017 Mar 1;64(5):666-674. doi: 10.1093/cid/ciw811. Epub 2016 Dec 10. PMID: 27940946.
- Rutter WC, Cox JN, Martin CA, Burgess DR, Burgess DS. Nephrotoxicity during Vancomycin Therapy in Combination with Piperacillin-Tazobactam or Cefepime. Antimicrob Agents Chemother. 2017 Jan 24;61(2):e02089-16. doi: 10.1128/AAC.02089-16. Erratum in: Antimicrob Agents Chemother. 2017 Mar 24;61(4): PMID: 27895019; PMCID: PMC5278703.
- Peyko V, Smalley S, Cohen H. Prospective Comparison of Acute Kidney Injury During Treatment With the Combination of Piperacillin-Tazobactam and Vancomycin Versus the Combination of Cefepime or Meropenem and Vancomycin. J Pharm Pract. 2017 Apr;30(2):209-213. doi: 10.1177/0897190016628960.
- Rutter WC, Burgess DS. Acute Kidney Injury in Patients Treated with IV Beta-Lactam/Beta-Lactamase Inhibitor Combinations. Pharmacotherapy. 2017 May;37(5):593-598. doi: 10.1002/phar.1918.
- Jeon N, Staley B, Klinker KP, Hincapie Castillo J, Winterstein AG. Acute kidney injury risk associated with piperacillin/tazobactam compared with cefepime during vancomycin therapy in hospitalised patients: a cohort study stratified by baseline kidney function. Int J Antimicrob Agents. 2017 Jul;50(1):63-67. doi: 10.1016/j.ijantimicag.2017.02.023.
- Mousavi M, Zapolskaya T, Scipione MR, Louie E, Papadopoulos J, Dubrovskaya Y. Comparison of Rates of Nephrotoxicity Associated with Vancomycin in Combination with Piperacillin-Tazobactam Administered as an Extended versus Standard Infusion. Pharmacotherapy. 2017 Mar;37(3):379-385. doi: 10.1002/phar.1901. E
- Miano TA, Hennessy S, Yang W, Dunn TG, Weisman AR, Oniyide O, Agyekum RS, Turner AP, Ittner CAG, Anderson BJ, Wilson FP, Townsend R, Reilly JP, Giannini HM, Cosgriff CV, Jones TK, Meyer NJ, Shashaty MGS. Association of vancomycin plus piperacillin-tazobactam with early changes in creatinine versus cystatin C in critically ill adults: a prospective cohort study. Intensive Care Med. 2022 Sep;48(9):1144-1155. doi: 10.1007/s00134-022-06811-0.
- Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, Machado FR, Mcintyre L, Ostermann M, Prescott HC, Schorr C, Simpson S, Wiersinga WJ, Alshamsi F, Angus DC, Arabi Y, Azevedo L, Beale R, Beilman G, Belley-Cote E, Burry L, Cecconi M, Centofanti J, Coz Yataco A, De Waele J, Dellinger RP, Doi K, Du B, Estenssoro E, Ferrer R, Gomersall C, Hodgson C, Møller MH, Iwashyna T, Jacob S, Kleinpell R, Klompas M, Koh Y, Kumar A, Kwizera A, Lobo S, Masur H, McGloughlin S, Mehta S, Mehta Y, Mer M, Nunnally M, Oczkowski S, Osborn T, Papathanassoglou E, Perner A, Puskarich M, Roberts J, Schweickert W, Seckel M, Sevransky J, Sprung CL, Welte T, Zimmerman J, Levy M. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021 Nov;47(11):1181-1247. doi: 10.1007/s00134-021-06506-y.
- Qian ET, Casey JD, Wright A, Wang L, Shotwell MS, Siemann JK, Dear ML, Stollings JL, Lloyd BD, Marvi TK, Seitz KP, Nelson GE, Wright PW, Siew ED, Dennis BM, Wrenn JO, Andereck JW, Han JH, Self WH, Semler MW, Rice TW; Vanderbilt Center for Learning Healthcare and the Pragmatic Critical Care Research Group. Cefepime vs Piperacillin-Tazobactam in Adults Hospitalized With Acute Infection: The ACORN Randomized Clinical Trial. JAMA. 2023 Oct 24;330(16):1557-1567. doi: 10.1001/jama.2023.20583. PMID: 37837651; PMCID: PMC10576861.
Penicillin Allergy Cross Reactivity
Introduction
- Only 0.5% to 2% of patients with a documented penicillin allergy that are administered a penicillin will exhibit a hypersensitivity reaction, usually presenting as a rash or hives.
- True IgE-mediated penicillin allergies that cause anaphylaxis are rare.
- An IgE-mediated penicillin allergy can diminish over time, as 80% of patients become tolerant after a decade.
- Patients with a documented penicillin allergy may be inappropriately exposed to alternative antibiotics, resulting in increased treatment failures, adverse effects, and antimicrobial resistance.
- Penicillins, cephalosporins, and carbapenems all share a beta-lactam core structure, thus raising the potential for cross-reactivity among these agents.
Pharmacology
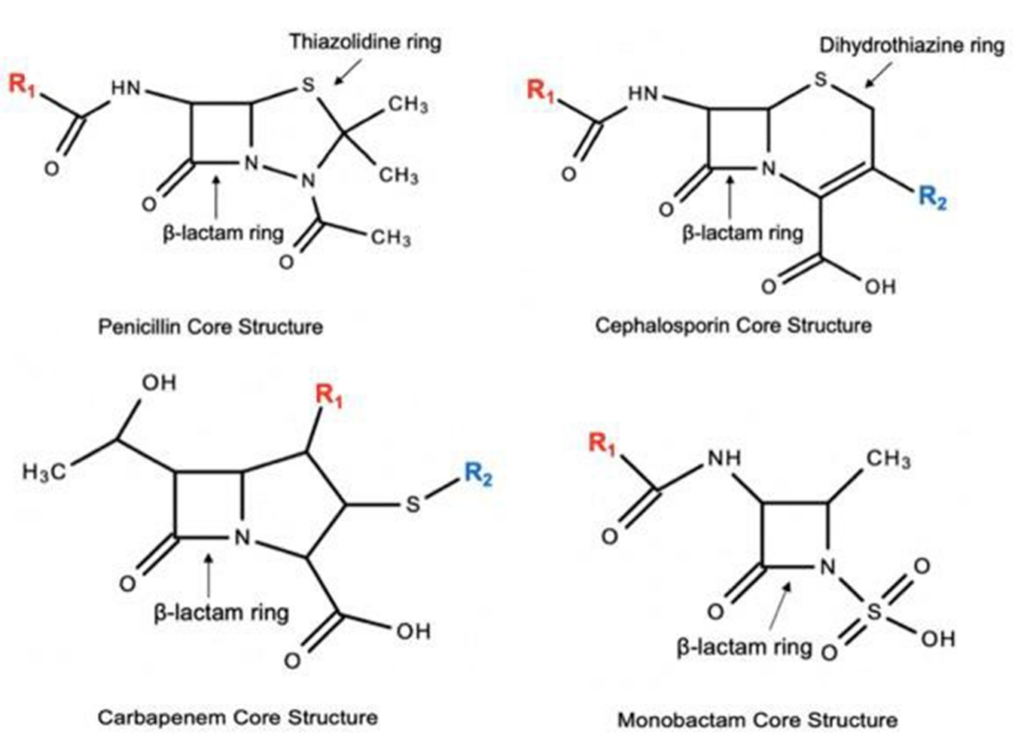
- The following drugs in each group may have cross-reactivity to each other due to similar side chains
- Cross-reactivity between penicillins and cephalosporins is about 2%
- Cefazolin is NOT likely to cross react with penicillin (side chains NOT similar)
- Cross-reactivity with monobactams (i.e. aztreonam) is negligible
- Cross-reactivity between penicillins and carbapenems is <1%
| Group 1 | Group 2 | Group 3 | Group 4 |
| Penicillin Cefoxitin Cefuroxime | Amoxicillin Ampicillin Cefaclor Cephalexin Cefadroxil | Ceftriaxone Cefotaxime Cefuroxime Cefepime Cefpodoxime Ceftaroline | Aztreonam Ceftolazane Ceftazidime |
Overview of Evidence
| Author | Design | Intervention & Comparison | Outcome | |
| Why Cross-Reactivity? | ||||
| Nagakura, 1990 Mayorga, 1995 | Animal study | -Studied antibodies formed when animals were immunized with protein-beta-lactam conjugates | -92% of the antibodies recognized an epitope in which the side chain was the main constituent -The side chain is the most important determinant in penicillin immunogenicity | |
| Cephalosporins | ||||
| Goodman, 2001 | Retrospective review (n=2933) | -Orthopedic patients with penicillin allergy receiving cefazolin prior to a procedure | Only 1 patient may have had an allergic reaction to cefazolinCross-reactivity rate with cefazolin was 0.33% | |
| Daulat, 2004 | Retrospective review (n=606) | -Patients with penicillin allergy receiving cephalosporins -42% 1st gen., 21% 2nd gen., and 37% 3rd or 4th gen. cephalosporins | Only 1 patient had an allergic reaction that was documented as worsening of underlying eczema after being placed on cefazolinCross-reactivity was 0.17% | |
| Apter, 2006 | Retrospective review (n=3920) | -Patients with a prescription for penicillin followed by a prescription for a cephalosporin -Identified allergic-like events within 30 days after each prescription | Only 43 patients who experienced an allergic- like reaction after both penicillin and cephalosporinCross-reactivity rate was 1.1%70% of these patients just had urticariaThe risk of anaphylaxis to cephalosporins was only 0.001% | |
| Romano, 2018 | Prospective review (n=252) | Prospective study of 252 subjects with IgE-mediated hypersensitivity to penicillins – Serum specific IgE assays for cefaclor and skin tests for 10 cephalosporins -Oral challenges with cefuroxime axetil, ceftriaxone, cefaclor, and cefadroxil for subjects with negative skin tests | 99 subjects (39.3%) had positive allergy tests for cephalosporins 95 subjects (37.7%) were positive to aminocephalosporins and/or cefamandole, which share side chains with penicillins All 244 subjects who underwent challenges with cefuroxime axetil and ceftriaxone tolerated them 7 subjects reacted to cefaclor or cefadroxil | |
| Carbapenems | ||||
| Romano, 2006 | Prospective study (n=112) | -Skin tested to penicillins and then skin tested to imipenem -If skin test to imipenem was negative, then challenged with IM dose | Only 1 patient of the penicillin skin-test positive patients had a positive skin test to imipenemCross-reactivity rate was 0.9%None of the 110 patients with a negative imipenem skin test that underwent IM challenge had a reaction | |
| Romano, 2007 | Prospective study (n=104) | -Skin tested to penicillins and then skin tested to meropenem -If skin test to imipenem was negative, then challenged with IV dose | Only 1 patient of the penicillin skin-test positive patients had a positive skin test to meropenemCross-reactivity rate was 1%All 103 patients with a negative meropenem skin test tolerated the IV challenge | |
| Atanaskovic- Markovic, 2008 | Prospective study (n=108) | -Children with penicillin allergy were skin tested to penicillin and meropenem -If skin test to meropenem was negative, then challenged with IV dose | Only 1 patient with a positive penicillin test reacted to the meropenem skin testCross-reactivity rate was 0.9%All 107 patients with a negative meropenem skin test tolerated the IV challenge | |
| Sánchez de Vicente, 2020 | Prospective study (n=137) | Tolerance testing for cephalosporins and carbapenems in patients with confirmed penicillin allergy | 0/46 patients showed positive skin tests for imipenem. 0.79% (1/137) patients showed a positive skin test for cefuroxime.0.79% (1/137) patients showed a positive skin test for ceftriaxone. | |
Conclusions
- True penicillin allergies are less common than reported, and anaphylaxis is uncommon.
- Cross-reactivity among penicillins and cephalosporins is attributed to similarity in side chains.
- Cephalosporin cross-reactivity with penicillins is much lower than reported in early studies partly due to contamination of study drugs with penicillin.
- Cross-reactivity between cephalosporins is about 2% and with carbapenems is <1%
References
- Apter AJ, Kinman JL, Bilker WB, et al. Is There Cross-Reactivity Between Penicillins and Cephalosporins? Am J Med. 2006;119(4):354e11-19.
- Atanaskovic-Markovic M, Gaeta F, Medjo B, Viola M, Nestorovic B, Romano A. Tolerability of Meropenem in Children with IgE-Mediated Hypersensitivity to Penicillins. Allergy. 2008;63:237-240.
- Blumenthal KG, Shenoy ES, Wolfson AR, et al. Addressing Inpatient Beta-Lactam Allergies: A Multihospital Implementation. J Allergy Clin Immunol Pract. 2017;5(3):616-625.
- Blumenthal KG, Huebner EM, Fu X, et al. Risk-Based Pathway for Outpatient Penicillin Allergy Evaluations. J Allergy Clin Immunol Pract. 2019;7(7):2411-2414.
- Campagna JD, Bond MC, Schabelman E, Hayes BD. The Use of Cephalosporins in Penicillin-Allergic Patients: A Literature Review. J Emerg Med. 2012;42(5):612-620.
- Chaudry SB, Veve MP, Wagner JL. Cephalosporins: A Focus on Side Chains and Beta-Lactam Cross-Reactivity. Pharmacy. 2019;7:1-16.
- Daulat S, Solensky R, Earl HS, Casey W, Gruchalla RS. Safety of Cephalosporin Administration to Patients with Hstories of Penicillin Allergy. J Allergy Clin Immunol Pract. 2004;113(6):1220-1222.
- DePestel DD, Benninger MS, Danziger L, et al. Cephalosporin Use in Treatment of Patients with Penicillin Allergies. J Am Pharm Assoc. 2008;48:530-540.
- Goodman EJ, Morgan MJ, Johnson PA, Nichols BA, Denk N, Gold BB. Cephalosporins can be Given to Penicillin-Allergic Patients Who Do Not Exhibit an Anaphylactic Response. J Clin Anesth. 2001;13(8):561-564.
- Mayorga C, Obispo T, Jimeno L, et al. Epitope Mapping of Beta-Lactam Antibiotics with the Use of Monoclonal Antibodies. Toxicology. 1995;97:225-234.
- Nagakura N, Souma S, Shimizu T, Yanagihara Y. Anti-Ampicillin Monoclonal Antibodies and their Cross- Reactivities to Various Beta-Lactams. Br J Hosp Med. 1990;44:252-258.
- Romano A, Viola M, Gueant-Rodriguez RM, Gaeta F, Pettinato R, Gueant JL. Imipenem in Patients with Immediate Hypersensitivity to Penicillins. N Engl J Med. 2006;354:2835-2837.
- Romano A, Viola M, Gueant-Rodriguez RM, Gaeta F, Valluzzi R, Gueant JL. Brief Communication: Tolerability of Meropenem in Patients with IgE-Mediated Hypersensitivity to Penicillins. Ann Intern Med. 2007;146:266-269.
- Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and Management of Penicillin Allergy: A Review. JAMA. 2019;321(2):188-199.
- Sánchez de Vicente J, Gamboa P, García-Lirio E, Irazabal B, Jáuregui I, Martínez MD, Segurola A, Seras Y, Galán C. Tolerance to Cephalosporins and Carbapenems in Penicillin-Allergic Patients. J Investig Allergol Clin Immunol. 2020;30(1):75-76. doi: 10.18176/jiaci.0463. Epub 2019 Nov 4. PMID: 31680067.
- Romano A, Valluzzi RL, Caruso C, Maggioletti M, Quaratino D, Gaeta F. Cross-Reactivity and Tolerability of Cephalosporins in Patients with IgE-Mediated Hypersensitivity to Penicillins. J Allergy Clin Immunol Pract. 2018 Sep-Oct;6(5):1662-1672. doi: 10.1016/j.jaip.2018.01.020. Epub 2018 Feb 3. PMID: 29408440.
Corticosteroids in Sepsis by Marissa Marks, PharmD
Introduction
- Sepsis is a systemic inflammatory response (SIRS) with associated organ dysfunction as a result of an infection.
- Sepsis is defined as ≥2 of the criteria:
- Temperature >38 ºC or <36 ºC
- Heart rate of >90 bpm
- Respiratory rate of >20 breaths/minute or pCO2 of <32 mmHg
- WBC >12,000 cells/mL or <4000 cells/mL
- Initial management of sepsis includes:
- Intravenous fluids (LR/NS) 30 mL/kg (based on total body weight) administered within the first 3 hours.
- Empiric antibiotic therapy based on the common bacteria and site of infection initiated within the first hour.
- Per the Surviving Sepsis guidelines, IV hydrocortisone is recommended for patients at least 4 hours after initiation of norepinephrine/epinephrine ≥0.25 mcg/kg/min to maintain a MAP of ≥65 mmHg.
Pharmacology
| Hydrocortisone | Methylprednisolone | Fludrocortisone | |
| Dose | IV: 50 mg Q6H or 100 mg Q8H x 5-7 days | IV (succinate): 40 to 125 mg/day (maximum of 1 to 2 mg/kg/day) | PO (in addition to another glucocorticoid): 0.05 mg/day x 7 days |
| Administration | IV: over ≥30 seconds | IV: over several minutes or over 15 to 60 minutes as an infusion | Administer by NG tube |
| PK/PD | -Onset of action (IV): 1 hour -T ½ elimination (IV): 2 +/- 0.3 hours | -Onset of action (IV): 1 hour -T ½ elimination (IV): 0.25 +/- 0.1 hour | -Onset of action (PO): 1-2 hours -T ½ elimination (PO): ~3.5 hours |
| Mechanism of Action | -Anti-inflammatory (decreased synthesis and release of inflammatory mediators) -Immunosuppressive (decreased response to hypersensitivity reactions) -Antiproliferative: vasoconstriction and decreased permeability of WBC to the injury | -Same mechanism of action as hydrocortisone with a 4-5x greater potency | – Mineralocorticoid activity > hydrocortisone or methylprednisolone |
| Adverse Effects | -Cardiovascular: increased blood pressure -Endocrine: fluid retention, hyperglycemia, weight gain -Gastrointestinal: increased appetite -Psychiatric: altered behavior | -Similar adverse effects as hydrocortisone | -Higher risk of fluid retention, hypertension, and decreased electrolyte concentrations |
| Drug Interactions and warnings | –Warnings: adrenal suppression, immunosuppression (higher doses for increased duration of therapy), psychiatric changes -Drug Interactions: antacids (separate by 2 hours), live vaccinations, DDAVP (risk of hyponatremia), succinylcholine | -Warnings: adrenal suppression, acute hepatitis (rare) -Drug Interactions: similar to hydrocortisone and fludrocortisone | -Warnings: patients with underlying hepatic dysfunction, myasthenia gravis, systemic sclerosis, or thyroid disease -Drug Interactions: similar to hydrocortisone and methylprednisolone |
| Compatibility | Drug in Solution: None tested | Drug in Solution: -Compatible: D5W- ½ NS, NS -Incompatible: D5W, D5NS, LR | N/A |
Overview of Evidence
| Author, year | Design/ sample size | Intervention & Comparison | Outcome | |
| French Trial Annane D, 2002. | RCT (n = 300) | hydrocortisone (50-mg intravenous bolus every 6 hours) and fludrocortisone (50- micro g tablet once daily) (n = 151) or matching placebos (n = 149) for 7 days. | 7-day treatment with low doses of hydrocortisone and fludrocortisone significantly reduced the risk of death in patients with septic shock and relative adrenal insufficiency without increasing adverse events. | |
| Teng-Jen Yu, 2009. | RCT (n = 40) | Hydrocortisone 50 mg IV Q6H or methylprednisolone 20 mg Q12H x 7 days | -Higher survival rates with hydrocortisone vs methylprednisolone | |
| VANISH Gordan, 2016 | RCT (n = 1400) | Vasopressin vs. norepinephrine plus hydrocortisone vs. placebo | No significant difference in mortality at 28 days, but vasopressin plus hydrocortisone was associated with faster reversal of shock and reduced need for renal replacement therapy | |
| Gibbison B, 2017. | Systematic review & meta-analysis (n = 33 clinical trials) | Systemic treatment with any corticosteroids | -Decreased septic shock reversal with methylprednisolone vs hydrocortisone -Increased 28-day mortality with methylprednisolone vs dexamethasone -Decreased risk of superinfections with methylprednisolone –Decreased ICU mortality and LOS with methylprednisolone | |
| CORTICUS Sprung, 2018 | RCT, (n=499) | Hydrocortisone 50 mg every 6 hours vs. placebo | The study found no significant difference between the two groups in 28-day mortality, but hydrocortisone was associated with a higher rate of shock reversal and a lower rate of progression to multiple organ dysfunction syndrome. | |
| HYPRESS Key, 2018 | RCT (n = 380) | Infusion of hydrocortisone 200 mg daily for five days followed by tapering until day 11 vs placebo | The study found no significant difference between the two groups in the primary outcome of time alive and free of vasopressor support by day 7 The study also found no significant difference between the two groups in secondary outcomes such as mortality at 28 days, ICU-free days, and hospital-free days | |
| ADRENAL Venkatesh B, 2018. | RCT (n = 3800) | Hydrocortisone 200 mg IV daily | -No difference in 28 or 90-day mortality with hydrocortisone –Decreased time to resolution of septic shock and discharge from the ICU with hydrocortisone -Decreased number of patients received a blood transfusion with hydrocortisone -Higher number of adverse events with hydrocortisone | |
| APROCCHHSAnnane D, 2018. | RCT (n = 1280) | -Hydrocortisone 50 mg IV Q6H + fludrocortisone 50 mcg PO daily in AM x 7 days -Drotrecogin alfa -Combination therapy of the three medications | –Decreased 90-day mortality with hydrocortisone + fludrocortisone -Decreased mortality with hydrocortisone + fludrocortisone at ICU and hospital discharge -Decreased time to discontinue vasopressor therapy and mechanical ventilation and achieve a SOFA score of <6 with hydrocortisone + fludrocortisone | |
Conclusions
- Per the Surviving Sepsis guidelines, hydrocortisone is recommended first-line for the treatment of septic shock in patients that are refractory to fluid (volume) resuscitation.
- Hydrocortisone portrayed greater efficacy in clinical trials than methylprednisolone.
- There are no clinical trials for the comparison of hydrocortisone monotherapy versus hydrocortisone + fludrocortisone; however, it is hypothesized that hydrocortisone provides sufficient mineralocorticoid activity as monotherapy without the increased risks of adverse effects with the addition of fludrocortisone.
- Necessary to avoid fludrocortisone in specific patient populations (i.e. congestive heart failure, hepatic and renal disease, etc.)
References
- Annane D, Buisson CB, Cariou A, Martin C, Misset B, Renault A, Lehmann B, Millul V, Maxime V, Bellissant E; APROCCHSS Investigators for the TRIGGERSEP Network. Design and conduct of the activated protein C and corticosteroids for human septic shock (APROCCHSS) trial. Ann Intensive Care. 2016 Dec;6(1):43.
- Annane D, Renault A, Brun-Buisson C, Megarbane B, Quenot JP, Siami S, Cariou A, Forceville X, Schwebel C, Martin C, Timsit JF, Misset B, Ali Benali M, Colin G, Souweine B, Asehnoune K, Mercier E, Chimot L, Charpentier C, François B, Boulain T, Petitpas F, Constantin JM, Dhonneur G, Baudin F, Combes A, Bohé J, Loriferne JF, Amathieu R, Cook F, Slama M, Leroy O, Capellier G, Dargent A, Hissem T, Maxime V, Bellissant E; CRICS-TRIGGERSEP Network. Hydrocortisone plus Fludrocortisone for Adults with Septic Shock. N Engl J Med. 2018 Mar 1;378(9):809-818.
- Evans L, Rhodes A, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021 Nov;47(11):1181-1247.
- Gibbison B, López-López JA, Higgins JP, Miller T, Angelini GD, Lightman SL, Annane D. Corticosteroids in septic shock: a systematic review and network meta-analysis. Crit Care. 2017 Mar 28;21(1):78.
- Hotchkiss RS, Moldawer LL, Opal SM, Reinhart K, Turnbull IR, Vincent JL. Sepsis and septic shock. Nat Rev Dis Primers. 2016 Jun 30;2:16045.
- Hydrocortisone (2023) UpToDate. Available at: https://www.uptodate.com (Accessed: 13 August 2023).
- Hydrocortisone Sodium Succinate (2023) Micromedex. Available at: https://www.micromedexsolutions.com (Accessed: 13 August 2023).
- Venkatesh B, Finfer S, Cohen J, Rajbhandari D, Arabi Y, Bellomo R, Billot L, Correa M, Glass P, Harward M, Joyce C, Li Q, McArthur C, Perner A, Rhodes A, Thompson K, Webb S, Myburgh J; ADRENAL Trial Investigators and the Australian–New Zealand Intensive Care Society Clinical Trials Group. Adjunctive Glucocorticoid Therapy in Patients with Septic Shock. N Engl J Med. 2018 Mar 1;378(9):797-808.
- Yu TJ, Liu YC, Yu CC, Tseng JC, Hua CC, Wu HP. Comparing hydrocortisone and methylprednisolone in patients with septic shock. Adv Ther. 2009 Jul;26(7):728-35.
- Keh D, Trips E, Marx G, Wirtz SP, Abduljawwad E, Bercker S, Bogatsch H, Briegel J, Engel C, Gerlach H, Goldmann A, Kuhn SO, Hüter L, Meier-Hellmann A, Nierhaus A, Kluge S, Lehmke J, Loeffler M, Oppert M, Resener K, Schädler D, Schuerholz T, Simon P, Weiler N, Weyland A, Reinhart K, Brunkhorst FM; SepNet–Critical Care Trials Group. Effect of Hydrocortisone on Development of Shock Among Patients With Severe Sepsis: The HYPRESS Randomized Clinical Trial. JAMA. 2016 Nov 1;316(17):1775-1785. doi: 10.1001/jama.2016.14799. PMID: 27695824.
- Sprung CL, Annane D, Keh D, Moreno R, Singer M, Freivogel K, Weiss YG, Benbenishty J, Kalenka A, Forst H, Laterre PF, Reinhart K, Cuthbertson BH, Payen D, Briegel J; CORTICUS Study Group. Hydrocortisone therapy for patients with septic shock. N Engl J Med. 2008 Jan 10;358(2):111-24. doi: 10.1056/NEJMoa071366. PMID: 18184957.
- Gordon AC, Mason AJ, Thirunavukkarasu N, Perkins GD, Cecconi M, Cepkova M, Pogson DG, Aya HD, Anjum A, Frazier GJ, Santhakumaran S, Ashby D, Brett SJ; VANISH Investigators. Effect of Early Vasopressin vs Norepinephrine on Kidney Failure in Patients With Septic Shock: The VANISH Randomized Clinical Trial. JAMA. 2016 Aug 2;316(5):509-18. doi: 10.1001/jama.2016.10485. PMID: 27483065.


