Author Archives: Jimmy
2020 American College of Rheumatology Guideline for the Management of Gout
GLOBAL STRATEGY FOR PREVENTION, DIAGNOSIS AND MANAGEMENT OF COPD: 2024 Report
2014 ESC Guidelines on the Diagnosis and Treatment of Aortic Diseases
Anaphylaxis 2
Statins for STEMI in the Emergency Department
Introduction
- STEMI (ST-Elevation Myocardial Infarction) represents a critical emergency where timely intervention is crucial. Atorvastatin, a statin, has been investigated for its potential benefits when administered early during a STEMI.
- Early administration of atorvastatin may have pleiotropic effects beyond cholesterol lowering. Potential benefits include stabilization of atherosclerotic plaques, reduction of inflammation, and improved endothelial function.
- Guidelines recommend initiating high-intensity statin therapy as soon as possible in STEMI patients.
- This pharmacy pearl summarizes the pharmacology and evidence supporting the use of atorvastatin in this setting.
Pharmacology
| Atorvastatin | Rosuvastatin | |
| Dose | 80 mg orally once daily | 40 mg orally once daily |
| Administration | Oral | Oral |
| PK/PD | Onset: 3-5 days for LDL reduction; Peak effect: 2-4 weeks | Onset: 3-5 days for LDL reduction; Peak effect: 2-4 weeks |
| Adverse Effects | Myopathy, elevated liver enzymes, gastrointestinal symptoms | Myopathy, elevated liver enzymes, gastrointestinal symptoms |
| Drug Interactions and warnings | CYP3A4 inhibitors/inducers can affect levels; avoid in active liver disease | Minimal CYP interactions; avoid in active liver disease |
| Compatibility | Compatible with most cardiovascular drugs, monitor for interactions with CYP3A4 inhibitors | Compatible with most cardiovascular drugs, minimal interactions |
| Comments | High-intensity statin recommended post-STEMI to reduce recurrence risk | High-intensity statin alternative to atorvastatin |
Overview of Evidence
| Author, Year | Design/Sample Size | Intervention & Comparison | Outcome | |
| Schwartz, 2001 | Randomized Controlled Trial (n=3086) | Atorvastatin (80 mg/day) vs. placebo initiated 2496 hours after acute coronary syndrome | Atorvastatin reduced recurrent symptomatic ischemia requiring rehospitalization (6.2% vs 8.4%; RR, 0.74; P=0.02) | |
| Li, 2012 | Randomized Controlled Trial (n=161) | High-dose atorvastatin (80 mg) vs. placebo in patients with STEMI undergoing PCI | High-dose atorvastatin significantly reduced the incidence of contrast-induced nephropathy (2.6% vs 15.7%; P=0.01) | |
| Liu, 2013 | Randomized Controlled Trial (n=102) | Loading dose of atorvastatin (80 mg) before PCI vs. no loading dose | Loading dose of atorvastatin reduced high-sensitivity C-reactive protein, B-type natriuretic peptide, and matrix metalloproteinase type 9, indicating reduced inflammation and improved cardiac function (P<0.05) | |
| Xu, 2016 | Randomized Controlled Trial (n=120) | Intensive atorvastatin (40 mg) vs. standard atorvastatin (20 mg) in STEMI patients undergoing PCI | Intensive atorvastatin significantly reduced serum endothelin-1 levels and ADP-induced platelet clot strength, improving endothelial function and platelet inhibition (P<0.05) | |
| Kim, 2015 | Randomized Controlled Trial (n=67) | High-dose atorvastatin (80 mg) before PCI vs. low-dose atorvastatin (10 mg) | No significant reduction in myocardial damage; however, high-dose pretreatment is generally considered safe and well-tolerated | |
| Gavazzoni, 2017 | Randomized Controlled Trial (n=52) | High-dose atorvastatin (80 mg) vs. moderate dose (20 mg) in STEMI patients | High-dose atorvastatin showed significant improvement in endothelial function (RH-PAT index 1.96±0.16 vs 1.72±0.19; P=0.002) and reduced levels of high-sensitivity CRP and IL6 (P<0.05) | |
| Liu, 2013 | Randomized Controlled Trial (n=102) | Loading dose of atorvastatin (80 mg) before PCI vs. no loading dose | Loading dose of atorvastatin significantly lowered inflammatory markers and improved left ventricular ejection fraction compared to no loading dose (P<0.05) | |
| Adel, 2022 | Randomized Controlled Trial (n=99) | High-dose rosuvastatin (40 mg) vs. high-dose atorvastatin (80 mg) before PCI in STEMI patients | Atorvastatin group had lower CTFC and better TIMI flow grade compared to control, and both statins improved microvascular myocardial perfusion (P<0.01) | |
| Chen, 2022 | Randomized Controlled Trial (n=98) | Enhanced-dose atorvastatin (40 mg before PCI, 40 mg/day post-PCI, 20 mg/day after 1 week) vs. standarddose atorvastatin (20 mg/day) | Enhanced-dose atorvastatin improved cardiac output, LVEF, TIMI blood flow classification, and reduced incidence of major adverse cardiac events (P<0.05) | |
Conclusions
- Efficacy: High-intensity atorvastatin (80 mg) initiated early in the ED for STEMI patients reduces the risk of subsequent cardiovascular events and mortality.
- Safety: Generally well-tolerated with a similar side effect profile to other statins, though monitoring for myopathy and liver enzyme elevations is necessary.
- Recommendation: Incorporating early administration of atorvastatin 80 mg for STEMI patients in the ED aligns with current guidelines and improves patient outcomes.
References
- Micromedex [Electronic version].Greenwood Village, CO: Truven Health Analytics. Retrieved July 1 2024, from http://www.micromedexsolutions.com/
- Schwartz GG, Olsson AG, Ezekowitz MD, et al. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA. 2001;285(13):1711-1718.
- Liu H, Yang Y, Yang SL, et al. Administration of a loading dose of atorvastatin before emergency PCI reduces myocardial damage in patients with STEMI. Clin Ther. 2013;35(1):22-30.
- Li W, Fu X, Wang Y, et al. Beneficial effects of high-dose atorvastatin pretreatment on microvascular obstruction and left ventricular function in STEMI patients undergoing primary PCI. Cardiology. 2012;123(4):212-220.
- Kim EK, Hahn J, Song Y, et al. Effects of high-dose atorvastatin pretreatment on microvascular obstruction in STEMI patients undergoing primary PCI. J Korean Med Sci. 2015;30(4):435-441.
- Xu X, Liu Y, Li K, et al. Intensive atorvastatin improves endothelial function and reduces inflammation in STEMI patients undergoing primary PCI. Int J Cardiol. 2016;220:616-621.
- Gavazzoni M, Lombardi CM, Vizzardi E, et al. Role of early high-dose atorvastatin loading in STsegment elevation myocardial infarction: real-life experience. J Cardiovasc Med (Hagerstown). 2017;18(6):406-411.
- Adel EM, Elberry A, Abdel Aziz A, Ibrahim MA, Abdelaal FA. Comparison of the treatment efficacy of rosuvastatin versus atorvastatin in preventing microvascular obstruction in patients undergoing primary PCI for STEMI. J Clin Med. 2022;11(17):5142.
- Chen Y, Zhang J, Huo Y, et al. Effects of atorvastatin on coronary microvascular function in STEMI patients undergoing primary PCI: a randomized controlled trial. J Am Coll Cardiol. 2022;79(9):901911.
Tranexamic Acid in Trauma by Jordan Spurling
Introduction
- Trauma is the leading cause of death in individuals up to 45 years old and the fourth leading cause of death overall for all ages.1
- Uncontrolled hemorrhage is the leading cause of early mortality in major trauma.2
- Trauma-associated hemorrhagic death occurs as an effect of uncontrolled bleeding and trauma-induced coagulopathy.3
- Tranexamic acid is an antifibrinolytic medication that works by forming a reversible complex that displaces plasminogen from fibrin resulting in inhibition of fibrinolysis.4
- Tranexamic acid is readily available, simple to administer, relatively inexpensive, with minimal side effects.
Pharmacology
| Tranexamic Acid | |
| Dose | Loading dose: 1 g over 10 minutes started within 3 hours of injury2 gram via slow IV Push* Maintenance: 1 g over the next 8 hours as a continuous infusion |
| Administration | Loading dose: administer undiluted Max rate:100 mg/minute For continuous IV infusions: dilute with compatible solutions and administer at a rate not to exceed 100 mg/minute |
| PK/PD | Distribution: Vd: IV: 9 to 12 L Protein binding: ~3%, primarily to plasminogen Half-life elimination: ~2 hours Excretion: Urine (>95% as unchanged drug) |
| Adverse Effects | Hypersensitivity reactions, ocular effects, seizures and myoclonus, thromboembolic effects, abdominal pain, headache, back pain |
Overview of Evidence
| Author, year | Design/ sample size | Intervention & Comparison | Outcome |
| Morrison, 2012 | ○ Observational (n=896) | ○ TXA 1g bolus + repeat prn vs placebo. | All-cause mortality overall within 48 hours and in hospital mortality significantly reduced with TXA |
| Roberts, 2013 | ○ Randomized placebo-controlled (n = 20,2011) | ○ TXA 1g bolus + 1g over 8 hours vs placebo | ○ All-cause mortality at 28 days was significantly reduced by TXA ○ Treatment within 1 hour and 1-3 hours from injury significantly reduced the risk of death due to bleeding |
| Sprigg, 2018 | ○ Randomized placebo-controlled (n= 2325) | ○ TXA 1 g bolus + 1g over 8 h infusion vs placebo | ○ Patients in the tranexamic acid group experienced a reduction in early deaths and serious adverse events, but not long term functional status |
| Roberts, 2019 | ○ Randomized, placebo-controlled (n=12737) | ○ TXA 1 g bolus + 1g over 8 hours vs placebo | ○ Treatment within 3 h of injury reduced head injury-related death. |
| Rowell, 2020 | ○ Double-blinded, randomized ( n= 966) | ● TXA 1 g bolus + 1 g 8-hour infusion vs 2 g bolus bolus + placebo infusion vs placebo bolus + placebo infusion | ○ No statistically significant difference in 28-day mortality, favorable neurologic function, 6 month disability rating score, or progression of intracranial hemorrhage |
| Roberts, 2020 | ○ Randomized placebo-controlled (n = 12,009) | ○ TXA 1 g + 3g infusion vs placebo | ○ No significant difference in death due to bleeding within 5 days |
| Bossers, 2021 | ● Prospective observational cohort (n = 1827) | ○ Pre-hospital TXA vs no TXA patients with TBI | ○ No association between TXA and mortality was found at 30 days ○ TXA was associated with increased mortality in patients with isolated TBI |
| Guyette, 2021 | ○ Double-blind, placebo-controlled, randomized ( n= 927) | ○ TXA 1 g bolus only vs TXA 1g + 1 g infusion vs TXA 1g bolus + 1g bolus + 1g infusion vs placebo bolus + placebo infusion | ○ Prehospital administration of tranexamic acid compared with placebo did not result in a lower rate of 30-day mortality in this population. ○ No differences were found in 24-hour mortality or in-hospital mortality |
| Mahmood, 2021 | ○ Randomized placebo-controlled (n = 1767) | ○ TXA 1 g bolus + 1 vs placebo | ○ No evidence that TXA prevents IPH expansion |
| Gruen, 2023 | ○ Double-blind, randomized, placebo-controlled (n = 1310) | ○ TXA 1 g bolus prior + infusion vs matched placebo | ○ No difference in survival with a favorable functional outcome at 6 months ○ No difference in 6 months mortality |
Conclusions
- Tranexamic acid has been studied in pre-hospital, hospital, and combat setting in patients who have sustained a traumatic injury
- Efficacy of tranexamic acid was demonstrated in some studies above, while other studies failed to show a significant difference in outcomes
- Dosing of tranexamic acid varied significantly in the above studies, however one dosing regimen has been widely adopted
- Tranexamic acid has minimal adverse effects, is relatively inexpensive, and readily available in many settings
References
- Rhee P, Joseph B, Pandit V, et al. Increasing trauma deaths in the United States. Ann Surg. 2014;260(1):13-21. doi:10.1097/SLA.0000000000000600
- Callcut RA, Kornblith LZ, Conroy AS, et al. The why and how our trauma patients die: A prospective Multicenter Western Trauma Association study. J Trauma Acute Care Surg. 2019;86(5):864-870. doi:10.1097/TA.0000000000002205
- Latif RK, Clifford SP, Baker JA, et al. Traumatic hemorrhage and chain of survival. Scand J Trauma Resusc Emerg Med. 2023;31(1):25. Published 2023 May 24. doi:10.1186/s13049-023-01088-8
- Hijazi N, Abu Fanne R, Abramovitch R, et al. Endogenous plasminogen activators mediate progressive intracerebral hemorrhage after traumatic brain injury in mice. Blood. 2015;125(16):2558-2567. doi:10.1182/blood-2014-08-588442
- Cai J, Ribkoff J, Olson S, et al. The many roles of tranexamic acid: An overview of the clinical indications for TXA in medical and surgical patients. Eur J Haematol. 2020;104(2):79-87. doi:10.1111/ejh.13348
- Morrison JJ, Dubose JJ, Rasmussen TE, Midwinter MJ. Military Application of Tranexamic Acid in Trauma Emergency Resuscitation (MATTERs) Study. Arch Surg. 2012;147(2):113-119. doi:10.1001/archsurg.2011.287
- Roberts I, Shakur H, Coats T, et al. The CRASH-2 trial: a randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess. 2013;17(10):1-79. doi:10.3310/hta17100
- Sprigg N, Flaherty K, Appleton JP, et al. Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial. Lancet. 2018;391(10135):2107-2115. doi:10.1016/S0140-6736(18)31033-X
- CRASH-3 trial collaborators. Effects of tranexamic acid on death, disability, vascular occlusive events and other morbidities in patients with acute traumatic brain injury
- Micromedex [Electronic version].Greenwood Village, CO: Truven Health Analytics. Retrieved January 17, 2021, from http://www.micromedexsolutions.com/
How to Fail the BCEMP Exam
The Board Certified Emergency Medicine Pharmacotherapy (BCEMP) exam, offered by the Board of Pharmacy Specialties, represents a significant milestone for healthcare professionals specializing in emergency medicine pharmacotherapy. This rigorous certification process is designed to validate a practitioner’s knowledge and skills, ensuring their capability to provide optimal medication therapy in emergency situations. While aiming for success is the natural course, today, we adopt a contrarian approach by highlighting the pitfalls that could lead to failure. This reverse-psychology method aims to illuminate the path towards effective preparation by understanding the common mistakes to avoid.
1. Misunderstanding the Exam’s Scope and Requirements
Understanding the full scope and requirements of the BCEMP exam is crucial for candidates aspiring to certify their expertise in emergency medicine pharmacotherapy. The Board of Pharmacy Specialties offers a detailed overview, including eligibility criteria, content areas, and a candidate’s guide on bpsweb.org, which serves as an essential resource for aligning preparation efforts with the exam’s demands. The exam encompasses a wide range of topics, from pharmacotherapy interventions to toxicological emergencies, requiring not just memorization but the ability to apply knowledge in complex clinical scenarios. Familiarity with these details ensures a targeted and effective study plan, addressing the depth of knowledge and breadth of topics expected.
Underestimating the breadth of topics covered in the BCEMP exam can lead to significant preparation gaps, potentially impacting exam performance. A narrow focus on certain areas while neglecting others can leave candidates ill-equipped to handle the diverse and complex questions posed. The comprehensive nature of the exam, designed to reflect the real-world challenges of emergency medicine, necessitates a thorough and well-rounded approach to study. By leveraging resources like the candidate’s guide from the Board of Pharmacy Specialties, candidates can avoid the pitfalls of underestimation and ensure a holistic preparation strategy that covers all necessary content areas.
2. Relying on Ineffective Study Techniques
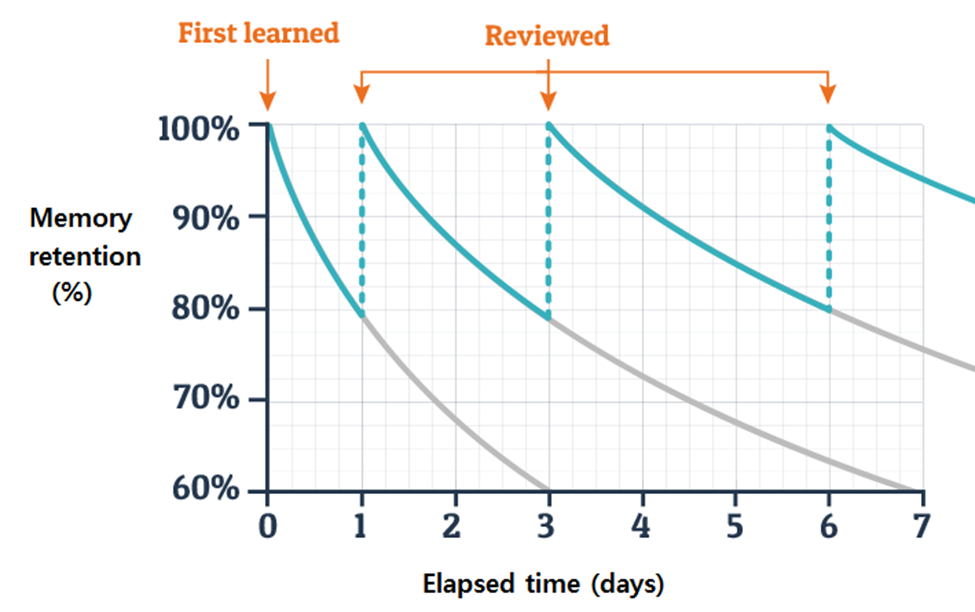
Relying solely on ineffective study techniques such as cramming and passive reading can be a major pitfall in preparing for the BCEMP exam. These methods are particularly unsuitable for mastering the complex and detailed information required in emergency medicine pharmacotherapy. Cramming, or the intense absorption of material shortly before the exam, fails to facilitate the deep learning necessary for long-term retention of intricate concepts. Passive reading, without active engagement, doesn’t reinforce the material in a meaningful way, making it difficult to recall under exam pressure. recall under exam pressure. The “Eight Recommendations to Promote Effective Study Habits for Biology Students Enrolled in Online Courses” emphasize the value of active learning, even in online
settings, suggesting that techniques fostering active engagement lead to more effective study habits. On the other hand, the benefits of spaced repetition, a study technique that involves reviewing material over increasing intervals, cannot be overstated. This method is grounded in the psychological principle of the spacing effect, which suggests that information is more easily recalled if learning sessions are spaced out over time. The Medium article “Importance of Spaced-Repetitions” highlights that spaced repetition significantly enhances long-term retention compared to massed practice (cramming), making it a superior strategy for complex subjects like pharmacotherapy. Additionally, the concept of the Ebbinghaus Forgetting Curve, detailed in “Ebbinghaus Forgetting Curve (Definition + Examples),” illustrates the natural decline of memory retention over time and how spaced repetition can counteract this effect. By incorporating these evidenced-based strategies into their study plan, candidates can greatly improve their grasp and retention of the extensive material covered in the BCEMP exam.
3. Overlooking Practice Exams and Question Banks
Overlooking the value of practice exams and question banks can be a critical misstep in preparing for the BCEMP exam. These tools are essential for familiarizing oneself with the exam’s structure and the variety of questions that may be encountered. A lack of exposure to the exam format and question types can lead to difficulties in navigating the actual test, potentially impacting performance. Incorporating practice materials into one’s study routine not only aids in understanding the exam’s layout but also helps in identifying weak areas that require additional focus.
Integrating practice exams and questions into the study schedule early and consistently is key to building confidence and competence. Engaging with a variety of questions, similar to those found in well-curated resources like PACUPrep’s Q-Bank, allows candidates to simulate the exam experience, enhancing their ability to manage time and approach questions effectively. This regular practice helps in cementing knowledge and honing test-taking strategies, making the actual exam environment more familiar and less intimidating. Through this disciplined approach, candidates can significantly improve their preparedness, paving the way for success in the BCEMP exam.
4. Poor Time Management
Poor time management in the context of BCEMP exam preparation can create significant barriers to success, primarily through the development of gaps in knowledge. Without a structured approach to studying, candidates risk spending too much time on certain topics while neglecting others, leading to an uneven grasp of the required material. This imbalance can be detrimental on exam day, where a comprehensive understanding across all areas is essential. Effective allocation of study time is crucial, ensuring that each topic is given due attention based on its complexity and the candidate’s familiarity with it.
To counteract this, adopting time management strategies can greatly enhance study efficiency. For instance, the Pomodoro Technique can be particularly effective in maintaining focus and preventing burnout. By dividing study sessions into 25-minute focused intervals followed by short breaks, candidates can improve retention and sustain longer periods of productive study. Additionally, the use of calendar apps to schedule study sessions and track important deadlines ensures a structured approach, covering all necessary topics systematically. During the exam, strategies such as quickly scanning all questions to allocate time effectively and keeping track of time to pace oneself can further optimize performance, ensuring that all questions are addressed within the allotted time.
5. Neglecting Mental and Physical Health
Neglecting mental and physical health during the preparation for the BCEMP exam can significantly undermine a candidate’s cognitive function and overall performance. The intense demands of studying for such a rigorous exam can lead to stress and sleep deprivation, which are known to impair memory, concentration, and decision-making abilities. Maintaining a healthy lifestyle, therefore, becomes not just a matter of personal well-being but a strategic component of exam preparation.
Adequate sleep is fundamental to cognitive processes, including the consolidation of memory, which is crucial when absorbing vast amounts of information. Similarly, effective stress management techniques such as regular exercise, mindfulness practices, or engaging in hobbies can mitigate the adverse effects of prolonged study sessions. These practices not only help in maintaining mental clarity and focus but also contribute to a more resilient and adaptable mindset, enabling candidates to tackle the challenges of the exam with greater composure and confidence. By prioritizing health and well-being, candidates can optimize their cognitive function, enhancing their ability to learn, retain, and apply knowledge effectively.
6. Studying in Isolation
Studying in isolation for the BCEMP exam can limit a candidate’s perspective and understanding, as it restricts the exchange of ideas, knowledge, and experiences that are vital for a comprehensive learning process. Engaging solely with the material without external input or discussion can lead to a narrow interpretation of complex topics and potentially overlook areas that might be crucial for the exam. The collaborative learning that occurs in group settings can expose candidates to diverse problem-solving approaches, clarify doubts through discussion, and reinforce learning through teaching others. To counteract the drawbacks of solitary study, candidates are encouraged to participate in study groups, professional forums, and seek mentorship.
7. Not Staying Updated with Current Practices
The field of emergency medicine pharmacotherapy is dynamic, with continuous advancements in treatment protocols, medication management, and clinical guidelines. Not incorporating the latest research findings, guidelines, and best practices into study materials for the BCEMP exam can lead to a knowledge base that is outdated and not reflective of current standards of care. This gap can be particularly detrimental in an exam setting that evaluates a candidate’s ability to apply contemporary, evidence-based practices in emergency medicine scenarios.
To ensure that study materials are current and comprehensive, candidates should regularly consult reputable medical journals in the field of emergency medicine and pharmacotherapy. These journals often publish the latest research, reviews, and consensus guidelines that can inform and update a candidate’s knowledge. Additionally, engaging in continuing education courses and participating in professional networks and associations can provide insights into emerging trends and consensus opinions in the field. These resources not only aid in preparing for the BCEMP exam but also contribute to a practitioner’s ongoing professional development, ensuring that their practice remains aligned with the highest standards of patient care in emergency medicine pharmacotherapy.
Conclusion: Embracing a Proactive and Balanced Approach to Exam Preparation
In preparing for the BCEMP exam, it’s crucial to avoid common pitfalls that can hinder success. Understanding the exam’s scope, avoiding ineffective study techniques, utilizing practice exams, managing time effectively, maintaining health, engaging with peers and mentors, and staying updated with current practices are all key to a well-rounded preparation strategy. Neglecting any of these areas can lead to gaps in knowledge, unnecessary stress, and ultimately, underperformance in the exam.
Embracing a proactive and balanced approach to studying is essential. This means not only covering all the necessary material but doing so in a way that promotes deep understanding and long-term retention. Active learning, effective time management, collaboration, and continuous learning are all part of a holistic study plan. The goal is not just to pass the BCEMP exam but to excel in the field of emergency medicine pharmacotherapy, ensuring that you are well-equipped to provide the highest level of care to patients in critical situations. By avoiding these common pitfalls and adopting a disciplined approach to preparation, candidates can position themselves for success, both in the exam and in their professional practice.
References
- American Board of Pharmacy Specialties. Pharmacotherapy: Specialty Certification Examination Content Outline. https://www.bpsweb.org/wp-content/uploads/bps-specialties/pharmacotherapy/PT_contentoutline.pdf. Accessed September 28, 2021.
- Dunlosky J, Rawson KA, Marsh EJ, Nathan MJ, Willingham DT. Improving students’ learning with effective learning techniques: promising directions from cognitive and educational psychology. Psychol Sci Public Interest. 2013;14(1):4-58. doi:10.1177/1529100612453266
- Brown PC, Roediger HL 3rd, McDaniel MA. Make It Stick: The Science of Successful Learning. Cambridge, MA: Belknap Press of Harvard University Press; 2014.
- Johnson M, Taubenheim A, Samar H, et al. Examining the impact of practice testing on learning and retention of medical information. Curr Pharm Teach Learn. 2020;12(9):1037-1045. doi:10.1016/j.cptl.2020.06.005
- Hartwig MK, Dunlosky J. Study strategies of college students: are self-testing and scheduling related to achievement? Psychon Bull Rev. 2012;19(1):126-134. doi:10.3758/s13423-011-0181-y
- McDaniel MA, Agarwal PK, Huelser BJ, McDermott KB, Roediger HL 3rd. Test-enhanced learning in a middle school science classroom: the effects of quiz frequency and placement. J Educ Psychol. 2011;103(2):399-414. doi:10.1037/a0021782
- Kornell N, Bjork RA. The promise and perils of self-regulated study. Psychon Bull Rev. 2007;14(2):219-224. doi:10.3758/bf03194055
- Chew SL. How to Get the Most Out of Studying Video Series. http://www.samford.edu/how-to-study/. Published 2011. Accessed September 28, 2021.
- Tuckman BW, Rapport MD. A 16-session time management intervention for college students with attention deficit hyperactivity disorder. Behav Modif. 2019;43(4):527-559. doi:10.1177/0145445518777735
- Dyrbye LN, Shanafelt TD, Sinsky CA, et al. Burnout among health care professionals: a call to explore and address this underrecognized threat to safe, high-quality care. NAM Perspectives. 2017;7(7). doi:10.31478/201707b
- Pagnini F, Bercovitz KE, Phillips D. Calm education: meta-analysis of mindfulness and emotion regulation strategies for teachers and students. Int J Stress Manag. 2018;25(4):364-376. doi>10.1037/str0000104
- Cohen S, Janicki-Deverts D, Miller GE. Psychological stress and disease. JAMA. 2007;298(14):1685-1687. doi:10.1001/jama.298.14.1685
- Krupat E, Richards JB, Sullivan AM, Fleenor TJ Jr, Schwartzstein RM. Assessing the effectiveness of case-based collaborative learning via randomized controlled trial. Acad Med. 2016;91(5):723-729. doi:10.1097/ACM.0000000000001044
- Karpicke JD, Blunt JR. Retrieval practice produces more learning than elaborative studying with concept mapping. Science. 2011;331(6018):772-775. doi:10.1126/science.1199327
- Singh N. Habituation, Memory, Spaced-Repetitions, Massed-Repetition. LeMe’24 Blog Post 4. March 7, 2024. Available from: https://medium.com/@nishchay.singh.23510074/habituation-memory-spaced-repetitions-massed-repetition-7640d4595429
- Ewell SN, Cotner S, Drake AG, Fagbodun S, Google A, Robinson L, Soneral P, Ballen CJ. 2022. Eight Recommendations to Promote Effective Study Habits for Biology Students Enrolled in Online Courses. J Microbiol Biol Educ. 23:e00260-21.
PROPHY-VAP Trial
Study To Review
One Sentence Summary
The PROPHY-VAP trial showed that a single dose of ceftriaxone can significantly reduce early-onset VAP in mechanically ventilated brain-injured patients, offering a potential change in clinical practice for VAP prevention.
Introduction
Methods
Results
Ventilator-associated pneumonia (VAP) is a significant concern in intensive care units (ICU), especially prevalent among brain-injured patients who are mechanically ventilated. With VAP incidence rates ranging from 22% to 71%, depending on the study, and brain injury being a notable independent risk factor, the prevention of this infection is paramount. Current standard of care includes a set of measures aiming to reduce the incidence of VAP; however, the challenge of early versus late onset VAP and the associated risk of multi-drug resistant organisms necessitate ongoing research and innovation in prophylactic strategies. The PROPHY-VAP trial is a pivotal study that explores the use of prophylactic antibiotics, specifically ceftriaxone, to prevent early-onset VAP in brain-injured patients. By addressing the controversial cut-off for early VAP onset in this population and considering the unique susceptibility profile of their infections, this trial holds significant importance for enhancing patient outcomes and refining ICU protocols.
PROPHY-VAP was a multicentre, randomized, double-blind, placebo-controlled trial conducted in nine French ICUs. The study enrolled comatose adult patients (Glasgow Coma Scale score ≤12) requiring mechanical ventilation post-acute brain injury. Patients were randomly assigned to receive either intravenous ceftriaxone 2 g or a placebo within 12 hours of tracheal intubation. The primary outcome was the incidence of early VAP (2nd to 7th day of mechanical ventilation). The study excluded patients with high death risk within 48 hours, previous hospitalizations for coma, contraindications to beta-lactams, and those receiving antibiotics for pre-existing infections.
- The PROPHY-VAP trial was conducted over a period spanning from October 14, 2015, to May 27, 2020, during which 345 patients were randomized in a 1:1 ratio to receive either ceftriaxone (171 patients) or placebo (174 patients). The study observed a marked reduction in the incidence of early VAP in the ceftriaxone group, with only 14% (23 patients) developing the infection compared to 32% (51 patients) in the placebo group, translating to a hazard ratio of 0.60. This significant decrease in early-onset VAP incidence, demonstrated in a double-blind, placebo-controlled setting, suggests that a single dose of ceftriaxone can be an effective prophylactic intervention in brain-injured patients requiring mechanical ventilation.
A detailed analysis of the patient demographics within the trial revealed that out of the 319 patients included in the analysis, 166 were men and 153 were women. Adjudication confirmed a total of 93 cases of VAP, of which 74 were early infections. Importantly, the study reported no increase in multi-drug resistant organisms or adverse effects attributable to the administration of ceftriaxone. These results indicate that the intervention was not only efficacious in reducing the incidence of early VAP but also safe for the patients, with no additional risk of fostering antibiotic resistance. The authors report that these findings provide a strong argument for the inclusion of a single dose of ceftriaxone in VAP prevention bundles for this patient population.
Conclusion
The study demonstrates that a single dose of ceftriaxone significantly reduces the risk of early VAP in brain-injured patients requiring mechanical ventilation, without adverse microbiological consequences. This finding supports the inclusion of an early ceftriaxone dose in VAP prevention protocols for brain-injured patients.
Impression
The PROPHY-VAP trial’s findings have sparked debate within the medical community, with practitioners expressing both interest in the potential benefits of ceftriaxone prophylaxis and concern regarding the broader implications of antibiotic use, such as the risk of over-diagnosis of VAP and the absence of data on late-onset VAP and resistant pathogens. These discussions underscore the necessity for further research and careful consideration before integrating these results into standard practice.
Further Readings
Piperacillin-tazobactam plus Vancomycin and Acute Kidney Injury by Caroline Rosario
Introduction
- Vancomycin and piperacillin-tazobactam are combined for broad-spectrum antibiotic coverage including MRSA and Pseudomonas in hospitalized patients.
- AKI, often as acute tubular necrosis, is a known complication of vancomycin, especially with higher doses and co-administration of nephrotoxic drugs.
- Piperacillin-tazobactam alone has minimal nephrotoxicity (<1%); its nephrotoxicity is usually due to acute interstitial nephritis.
- Reported AKI rates vary in literature based on AKI definition and target population.
- Both drugs affect OAT1/3 transporters in the kidney, which are crucial for creatinine clearance and are especially significant in patients with CKD.
Pharmacology
| Vancomycin | Piperacillin-tazobactam4 | |
| Dose | Depends on infection and PK/PD target General dosing for systemic infections: IV 15-20 mg/kg IV Q8-12H for systemic infections | Standard infusion: 3.375 g IV Q6H over 30 minAntipseudomonal: 4.5 g IV Q6-8H over 30 minExtended infusion: 4.5 g IV then 3.375-4.5 g over 4 hours Q8H |
| Administration | Administer IV over ≥60 minutes at concentrations ≤5 mg/mL to reduce the risk of vancomycin infusion reaction | Standard infusion: Infuse over 30 min Extended infusion: Infuse loading dose over 30 min, start maintenance dose four hours later infused over 4 hours |
| PK/PD | Negligible oral bioavailability T1/2 = 4-6 hours Renally eliminated (40-100% unchanged) AUC:MIC dependent kinetics, PK/PD target AUC/MIC ≥400 µg/mL; surrogate serum trough concentrations often used | T1/2 = 0.7-1.2 hours Renally eliminated (80% unchanged) Dose adjust at CrCl<40 T>MIC dependent kinetics, prolonged infusions enhance efficacy |
| Adverse Effects | Nephrotoxicity Ototoxicity Vancomycin-infusion reaction (flushing, hypotension, tachycardia) | GI upset (diarrhea, nausea, constipation) Headache Rash, pruritis |
| Drug Interactions and warnings | Substrate of OAT1/3 +/- Inducer of OAT1/3 ↑ nephrotoxicity: aminoglycosides, aspirin | Piperacillin: substrate and inhibitor of OAT1/3∆, Tazobactam: substrate of OAT1/3 Interactions: Probenecid (↑ piperacillin-tazobactam), Methotrexate (↑ methotrexate) |
| Compatibility | Compatible with dextrose, NS, LR Incompatible with lipid emulsion | LR: only the formulation containing EDTA is compatible for Y-site administration Not chemically stable in solutions containing sodium bicarbonate or solutions that significantly alter pH Cannot be added to blood products or albumin hydrolysates |
| Comments | Serum troughs are a poor proxy of 24-hour AUC, trough-guided regimens have been shown to exceed the target AUC in 60% of adults10 | Useful in the ED for anaerobic coverage in Grade III open fractures, pneumonia with lung abscess or empyema, and empiric antipseudomonal coverage in patients with risk factors |
Overview of Evidence
| Author, year | Design/ sample size | Intervention & Comparison | AKI definition | Outcome |
| Sanz et al., 2002 | Prospective, multi-center (n = 969) | Amikacin+cefepime vs. amikacin+piperacillin-tazobactam | ↑ SCr ≥50% from baseline | No difference in severe nephrotoxicity between amikacin+piperacillin-tazobactam vs. amikacin+cefepime |
| Karino et al., 2016 | Retrospective cohort and nested case-control studies (n = 320) | Vancomycin+piperacillin-tazobactam standard infusion vs. Vancomycin+piperacillin-tazobactam extended-infusion | RIFILE criteriaAKIN criteriaVancomycin consensus guideline definition | AKI occurred in 33% of patients receiving vancomycin+piperacillin-tazobactamUse of extended infusion piperacillin-tazobactam did not increase risk of AKI Highest daily incidence of AKI occurred on day 5 of combination therapy |
| Hammond et al., 2017 | Meta-analysis of 14 observational studies (n = 3549) | Vancomycin+piperacillin-tazobactam vs. vancomycin+any β-lactam or vancomycin alone | All included studies used one of the following: RIFLE criteriaAKIN criteria↑ SCr ≥100% or >0.5 mg/dL | Vancomycin+piperacillin-tazobactam greater association with AKI (aOR, 3.11; 95% CI, 1.77–5.47) Highest incidence of AKI in patients admitted to the ICU (OR 3.83 95% CI, 1.67-8.78) |
| Rutter et al., 2017 | Retrospective matched cohort (n = 4103) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | RIFLE criteria | Vancomycin+piperacillin-tazobactam 2.18 times more likely to cause AKI vs. vancomycin+cefepime (95% CI, 1.64–2.94) Vancomycin doses between 3 and 4 g daily used, |
| Peyko et al., 2017 | Prospective observational cohort (n = 85) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime or vancomycin+meropenem | KDIGO | Incidence of AKI was higher in with vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime or meropenem (37.3% vs. 7.7% P = .005) |
| Rutter and Burgess et al., 2017 | Retrospective matched cohort (n = 2448) | Vancomycin+piperacillin-tazobactam vs. Vancomycin+ampicillin-sulbactam | RIFLE criteria | Increased risk of AKI with vancomycin+piperacillin-tazobactam (aOR, 1.77; 95% CI, 1.26–2.46), no increased rate of AKI with vancomycin+ampicillin-sulbactamRates of AKI similar for piperacillin-tazobactam and ampicillin-sulbactam without vancomycin |
| Jeon et al., 2017 | Retrospective matched cohort (n = 5335) | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | ↑ SCr ≥0.3 mg/dL or ≥50% from baseline | Vancomycin+piperacillin-tazobactam associated with a higher risk of AKI vs. vancomycin-cefepime (aHR, 1.25; 95% CI, 1.11–1.42.) |
| Mousavi et al., 2017 | Retrospective matched cohort (n = 280) | Vancomycin+piperacillin-tazobactam standard infusion vs. Vancomycin+piperacillin-tazobactam extended-infusion | RIFLE criteriaAKIN criteria | Similar rate of AKI between vancomycin+piperacillin-tazobactam standard infusion vs. vancomycin+piperacillin-tazobactam extended-infusionHigher vancomycin troughs were observed in the extended infusion group |
| Miano et al., 2022 | Prospective, observational | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime for ≥48 hours | ↑ SCr vs. ↑ Cystatin C vs. ↑ BUN | Vancomycin + piperacillin-tazobactam ➡️ ↑ serum creatinine-defined AKI, but no change in cystatin C, BUN, or AKI outcomes (dialysis/mortality).Indicates vancomycin + piperacillin-tazobactam AKI may be pseudotoxicity. |
| Qian et al, 2023 (ACORN Trial) | Randomized controlled Trial N=2511 | Vancomycin+piperacillin-tazobactam vs. vancomycin+cefepime | KDIGO ↑ SCr ≥0.3 mg/dL or ≥50% from baseline | The highest stage of acute kidney injury or death was not significantly different between the cefepime group and the piperacillin-tazobactam groupThe incidence of major adverse kidney events at day 14 did not differ between groups (124 patients [10.2%] in the cefepime group vs 114 patients [8.8%] in the piperacillintazobactam group~77% of each concurrently received vancomycin |
RIFLE, AKIN and KDIGO definitions of AKI are based upon ↑ in serum creatinine or ↓ in urine output
Conclusions
- Since 2011, evidence indicates combined vancomycin+ piperacillin-tazobactam may be nephrotoxic.
- Most studies were retrospective, defining nephrotoxicity by creatinine-based AKI.
- Recent data show this AKI definition doesn’t align with severe AKI outcomes (hemodialysis/mortality).
- Non-tubular secretion biomarkers (Cystatin C, BUN) didn’t show the same AKI increase.
- Despite >50 studies linking the drug combo with AKI, some expert report true renal risk is likely minimal.
- In emergencies, timely antibiotic use is vital; nephrotoxicity concerns shouldn’t delay this combo, especially for short use.
References
- Micromedex [Electronic version].Greenwood Village, CO: Truven Health Analytics. Retrieved October 4, 2023, from http://www.micromedexsolutions.com/
- VANCOMYCIN HYDROCHLORIDE [package insert]. Rockford, IL: Mylan Institutional LLC; Jul, 2018.
- Blair M, Côté JM, Cotter A, Lynch B, Redahan L, Murray PT. Nephrotoxicity from Vancomycin Combined with Piperacillin-Tazobactam: A Comprehensive Review. Am J Nephrol. 2021;52(2):85-97. doi: 10.1159/000513742.
- Pill MW, O’Neill CV, Chapman MM, Singh AK. Suspected acute interstitial nephritis induced by piperacillin-tazobactam. Pharmacotherapy. 1997 Jan-Feb;17(1):166-9..
- Li H, Yang Q, Gui M, Ding L, Yang L, Sun H, Li Z. Changes of renal transporters in the kinetic process of VCM-induced nephrotoxicity in mice. Toxicol Res (Camb). 2021 Jun 9;10(4):687-695. doi: 10.1093/toxres/tfab048. PMID: 34484661; PMCID: PMC8403606.
- Giuliano CA, Patel CR, Kale-Pradhan PB. Is the Combination of Piperacillin-Tazobactam and Vancomycin Associated with Development of Acute Kidney Injury? A Meta-analysis. Pharmacotherapy. 2016 Dec;36(12):1217-1228. doi: 10.1002/phar.1851.
- Boucher, H. (2023) Piperacillin-tazobactam, Sanford Guide Web Edition. Available at: https://webedition.sanfordguide.com/en/drug-information/antibacterial-agents/penicillins/anti-pseudomonal-penicillins/piperacillin-tazobactam (Accessed: 12 October 2023).
- Yang S, Liu Z, Wang C, Wen S, Meng Q, Huo X, Sun H, Ma X, Peng J, He Z, Liu K. Piperacillin enhances the inhibitory effect of tazobactam on β-lactamase through inhibition of organic anion transporter 1/3 in rats. Asian J Pharm Sci. 2019 Nov;14(6):677-686. doi: 10.1016/j.ajps.2018.11.003.
- Landersdorfer CB, Kirkpatrick CM, Kinzig M, Bulitta JB, Holzgrabe U, Sörgel F. Inhibition of flucloxacillin tubular renal secretion by piperacillin. Br J Clin Pharmacol. 2008 Nov;66(5):648-59. doi: 10.1111/j.1365-2125.2008.03266.x.
- Neely MN, Youn G, Jones B, Jelliffe RW, Drusano GL, Rodvold KA, Lodise TP. Are vancomycin trough concentrations adequate for optimal dosing? Antimicrob Agents Chemother. 2014;58(1):309-16. doi: 10.1128/AAC.01653-13.
- Alvarez-Arango S, Ogunwole SM, Sequist TD, Burk CM, Blumenthal KG. Vancomycin Infusion Reaction – Moving beyond “Red Man Syndrome”. N Engl J Med. 2021 Apr 8;384(14):1283-1286. doi: 10.1056/NEJMp2031891. Epub 2021 Apr 3.
- Vallon V, Eraly SA, Rao SR, Gerasimova M, Rose M, Nagle M, Anzai N, Smith T, Sharma K, Nigam SK, Rieg T. A role for the organic anion transporter OAT3 in renal creatinine secretion in mice. Am J Physiol Renal Physiol. 2012 May 15;302(10):F1293-9. doi: 10.1152/ajprenal.00013.2012. Epub 2012 Feb 15. PMID: 22338083; PMCID: PMC3362066.
- Sanz MA, López J, Lahuerta JJ, Rovira M, Batlle M, Pérez C, Vázquez L, Julià A, Palau J, Gutiérrez M, Capote FJ, Ramos F, Benlloch L, Larrea L, Jarque I; Spanish PETHEMA Group. Cefepime plus amikacin versus piperacillin-tazobactam plus amikacin for initial antibiotic therapy in haematology patients with febrile neutropenia: results of an open, randomized, multicentre trial. J Antimicrob Chemother. 2002 Jul;50(1):79-88. doi: 10.1093/jac/dkf087. PMID: 12096010.
- Watkins RR, Deresinski S. Increasing Evidence of the Nephrotoxicity of Piperacillin/Tazobactam and Vancomycin Combination Therapy-What Is the Clinician to Do? Clin Infect Dis. 2017 Nov 29;65(12):2137-2143. doi: 10.1093/cid/cix675.
- Karino S, Kaye KS, Navalkele B, Nishan B, Salim M, Solanki S, Pervaiz A, Tashtoush N, Shaikh H, Koppula S, Martin ET, Mynatt RP, Murray KP, Rybak MJ, Pogue JM. Epidemiology of Acute Kidney Injury among Patients Receiving Concomitant Vancomycin and Piperacillin-Tazobactam: Opportunities for Antimicrobial Stewardship. Antimicrob Agents Chemother. 2016 May 23;60(6):3743-50. doi: 10.1128/AAC.03011-15.
- Hammond DA, Smith MN, Li C, Hayes SM, Lusardi K, Bookstaver PB. Systematic Review and Meta-Analysis of Acute Kidney Injury Associated with Concomitant Vancomycin and Piperacillin/tazobactam. Clin Infect Dis. 2017 Mar 1;64(5):666-674. doi: 10.1093/cid/ciw811. Epub 2016 Dec 10. PMID: 27940946.
- Rutter WC, Cox JN, Martin CA, Burgess DR, Burgess DS. Nephrotoxicity during Vancomycin Therapy in Combination with Piperacillin-Tazobactam or Cefepime. Antimicrob Agents Chemother. 2017 Jan 24;61(2):e02089-16. doi: 10.1128/AAC.02089-16. Erratum in: Antimicrob Agents Chemother. 2017 Mar 24;61(4): PMID: 27895019; PMCID: PMC5278703.
- Peyko V, Smalley S, Cohen H. Prospective Comparison of Acute Kidney Injury During Treatment With the Combination of Piperacillin-Tazobactam and Vancomycin Versus the Combination of Cefepime or Meropenem and Vancomycin. J Pharm Pract. 2017 Apr;30(2):209-213. doi: 10.1177/0897190016628960.
- Rutter WC, Burgess DS. Acute Kidney Injury in Patients Treated with IV Beta-Lactam/Beta-Lactamase Inhibitor Combinations. Pharmacotherapy. 2017 May;37(5):593-598. doi: 10.1002/phar.1918.
- Jeon N, Staley B, Klinker KP, Hincapie Castillo J, Winterstein AG. Acute kidney injury risk associated with piperacillin/tazobactam compared with cefepime during vancomycin therapy in hospitalised patients: a cohort study stratified by baseline kidney function. Int J Antimicrob Agents. 2017 Jul;50(1):63-67. doi: 10.1016/j.ijantimicag.2017.02.023.
- Mousavi M, Zapolskaya T, Scipione MR, Louie E, Papadopoulos J, Dubrovskaya Y. Comparison of Rates of Nephrotoxicity Associated with Vancomycin in Combination with Piperacillin-Tazobactam Administered as an Extended versus Standard Infusion. Pharmacotherapy. 2017 Mar;37(3):379-385. doi: 10.1002/phar.1901. E
- Miano TA, Hennessy S, Yang W, Dunn TG, Weisman AR, Oniyide O, Agyekum RS, Turner AP, Ittner CAG, Anderson BJ, Wilson FP, Townsend R, Reilly JP, Giannini HM, Cosgriff CV, Jones TK, Meyer NJ, Shashaty MGS. Association of vancomycin plus piperacillin-tazobactam with early changes in creatinine versus cystatin C in critically ill adults: a prospective cohort study. Intensive Care Med. 2022 Sep;48(9):1144-1155. doi: 10.1007/s00134-022-06811-0.
- Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, Machado FR, Mcintyre L, Ostermann M, Prescott HC, Schorr C, Simpson S, Wiersinga WJ, Alshamsi F, Angus DC, Arabi Y, Azevedo L, Beale R, Beilman G, Belley-Cote E, Burry L, Cecconi M, Centofanti J, Coz Yataco A, De Waele J, Dellinger RP, Doi K, Du B, Estenssoro E, Ferrer R, Gomersall C, Hodgson C, Møller MH, Iwashyna T, Jacob S, Kleinpell R, Klompas M, Koh Y, Kumar A, Kwizera A, Lobo S, Masur H, McGloughlin S, Mehta S, Mehta Y, Mer M, Nunnally M, Oczkowski S, Osborn T, Papathanassoglou E, Perner A, Puskarich M, Roberts J, Schweickert W, Seckel M, Sevransky J, Sprung CL, Welte T, Zimmerman J, Levy M. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021 Nov;47(11):1181-1247. doi: 10.1007/s00134-021-06506-y.
- Qian ET, Casey JD, Wright A, Wang L, Shotwell MS, Siemann JK, Dear ML, Stollings JL, Lloyd BD, Marvi TK, Seitz KP, Nelson GE, Wright PW, Siew ED, Dennis BM, Wrenn JO, Andereck JW, Han JH, Self WH, Semler MW, Rice TW; Vanderbilt Center for Learning Healthcare and the Pragmatic Critical Care Research Group. Cefepime vs Piperacillin-Tazobactam in Adults Hospitalized With Acute Infection: The ACORN Randomized Clinical Trial. JAMA. 2023 Oct 24;330(16):1557-1567. doi: 10.1001/jama.2023.20583. PMID: 37837651; PMCID: PMC10576861.


